Frequently asked questions
What is hair regeneration?
Hair regeneration, or hair multiplication, is the formation of brand new hair follicles and is technically referred to as ‘follicle neogenesis’.
It has been proposed for many years as the ultimate treatment for hair loss. Hair transplantation successfully moves hairs from one region of the scalp to balding regions but is not able to increase the total number of hairs available.
Hair multiplication would, in effect, disassemble a few hair follicles, multiply these cells in the laboratory and then reintroduce them into the scalp to both rejuvenate miniaturising hair follicles and induce brand-new hairs. Other groups have tried this, but it has been found that when human follicle cells are cultured, they rapidly lose their functionality, and it is now generally accepted that hair multiplication/hair regeneration will require the formation of hair rudiments before implantation. Several groups are working actively in this area.
Follicle neogenesis on a scale that would be clinically useful faces considerable technical, regulatory, and financial hurdles, meaning that hair multiplication is unfortunately not a viable treatment option in the near future.
What is hair rejuvenation?
Hair rejuvenation is the process of rescuing failing hair follicles. In the context of androgenetic alopecia, hair rejuvenation targets actively miniaturising hair follicles in order to restore their original hair density.
Rejuvenating hair follicles has the potential to maintain hair density throughout life if hairs are treated early enough. The aim of this approach is to prevent hairs from fully miniaturising and to restore their original density. Alternatively, rejuvenation could be used in addition to hair transplantation, where some areas may have progressed too far, in order to gain an optimal result for patients.
Long-term hair rejuvenation represents a huge unmet need for treating androgenetic alopecia. Current options depend on prevention (e.g. finasteride), transient stimulation of follicles (e.g. PRP and minoxidil), or replacement (hair transplantation). Rejuvenating follicles using cell therapy could allow for a low-maintenance treatment that has long-lasting results.
What is Cryopreservation?
Cryopreservation is a process where biological material is preserved by cooling to very low temperatures (typically -196 °C). At low enough temperatures, any chemical activity in the cells is effectively stopped, and cells enter a state of ‘suspended animation’.
Methods of cryopreservation seek to reach low temperatures without the formation of damaging ice crystals during freezing. At these ultra-low temperatures, cells can be stored for many years and, if thawed correctly, are then able to resume normal function.
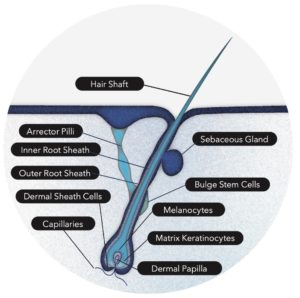
What are Dermal Papilla Cells?
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3115771/
The characteristics of DP cells also make them a source of induced pluripotent stem cells (iPSCs), as they require minimal reprogramming to be used as such.
What is Scientific Innovation
Classic scientific innovation develops potential new therapies often in isolation in the laboratory. The first time clinicians and patients are involved in any significant way is at the later stages of development during the clinical trial phase. By then the potential product specifics are determined, and the link with the scientists is broken.
This means that clinical trials often fail, not because the product doesn’t work, but because the trial design was incorrect. For example, the wrong patient group could have been tested, or the wrong procedure used to provide the product to the patient. This is much more critical with products that use surgery of some form or another, as it is the correct combination of product and surgery that is needed.
Unfortunately, when this is the case, the investment funds are not available to learn from the trial and start again. This has been the case with hair regeneration, and it has resulted in several companies trying to develop hair cloning and then moving out of the field entirely.
HairClone’s unique approach has involved a team of clinicians, scientists, and biotech entrepreneurs working together from the beginning. Early clinical testing is not done using a fixed protocol, allowing for potentially critical aspects of the product to be altered before completing international clinical trials. Samples from test subjects will be fed back to research scientists to gain in-depth molecular and cellular data to guide further treatments.
When will hair treatments be available
Our primary rejuvenation treatment requires the extraction of follicles, multiplication (cloning) of cells and injection back into the scalp. The initial step of extracting and storing follicles is already live, following HairClone’s launch of the World’s first follicle banking service. Patients can now bank follicles through one of our procurement centres. This allows immediate access to follicles when required.
The second stage involves multiplying dermal papilla cells. HairClone has built and furnished a Cell Manufacturing Facility in Guatemala City. The facility is situated a few floors below the Schambach Hair Clinic where treatments have begun testing. The clinical protocol will be undergoing optimisation over the coming months. During this time, local patients will be treated in order to complete rigorous follow-ups. Those willing to pay and travel to Guatemala for treatment will be considered on an individual basis following consultation with their local HairClone Clinical Partner (HairClone cannot offer medical advice). The treatment is still in its experimental phase and no assurances of outcome can be given.
In order to market our treatment, we must carry out clinical trials as soon as clinicians determine the most effective treatment system. It will take time to complete these trials and then apply for and obtain marketing licences from the regulatory bodies before this becomes a routine clinical treatment around the world; however, a small number of treatments could be continued at the Schambach Clinic.
HairClone’s management brings a huge amount of experience in this area. In previous positions, the team has carried out over 19 different clinical trials around the world and has developed a variety of cell therapies that have treated over 2 million people. We will do our utmost with the financial resources that we have to develop this therapy as quickly and efficiently as possible, but we will carry out each step ethically, with patient safety our top priority. Please keep checking the website and follow us on Instagram (@HairClone) to keep updated on our progress.
